|
|
| The degree of metamorphism*
is characterized by a mineral
assemblage* (see first page).
Minerals forming a metamorphic assemblage are by definition in equilibrium. This
equilibrium is characterized by various parameters notably rock and
minerals chemistry, presence of fluids, pressure and temperature.
This equilibrium generally corresponds to a maximum of temperature,
point called "metamorphic peak".
|
We apply on diagnostic
metamorphic assemblage the method known as "multi-equilibrium thermobarometry*".
On the basis of mineral chemistry, thermodynamic laws and database,
equilibria (or "reactions") can be computed and they should
intersect in one point, if equilibrium is achieved. |
Determination of Pressure-Temperature conditions in Lepontin Alps
(Engi, Todd & Schmatz 1995)
|
| More info on the web: TWQ
(R. Berman), Practical
aspect of Mineral Thermobarometry (D. J. Waters), THERMOCALC
(T. Holland and R. Powell) |
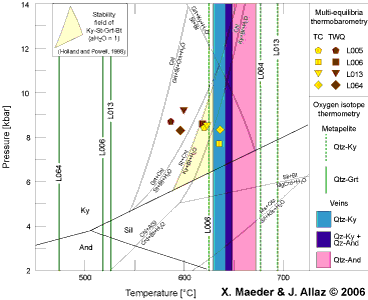
Pressure-Temperature results for micaschists of Alpe Larecc
And comparaison with temperatures estimated by oxygen isotops thermometry
(on mineral pair quartz-aluminosilicates). Errors not represented
on figure: maximum ±1 kbar, ±50°C
for TWQ and TC; ca. 50°C for oxygen isotope thermometry. |
The thermobarometric study has been done by comparison of two different
thermodynamic databases for mineral, THERMOCALC 3.1 (Holland
& Powell 1998 [reference] -
2002 [web])
and TWQ 2.02 (Berman 1988 [reference]
- 1997 [web]).
These two databases allowed a quantification of equilibrium (P and
T) in micaschist*
and paragneiss*
(all rocks are composed notably of garnet, staurolite and kyanite)
at about
620 to 650 °C and 8 to 10 kbar (= 0.8 to 1 GPa, 20 - 25
km depth). These conditions are coherent with the mineralogical assemblage
(triangle
of coexistence of garnet, staurolite, biotite and Kyanite in figure
on the left). The TWQ results show temperatures slightly lower (of
about
50°C), but comparable pressures.
|
The estimated
temperatures are slightly higher than those determined by Todd &
Engi (1997 [reference], figure
above). However, Todd and Engi have worked with the first version
of TWQ. Using this TWQ version with our analyses gives comparable
temperature but lower pressure (1-2 kb lower, results not presented
here).
|
|
Another method of
thermometry is based on the determination of oxygen isotopic ratio
(16 and 18, "d18O" method) of some
mineral couple to allow calculation of an equilibration temperature.
This method has been used on minerals of tension gashes (aluminosilicate-bearing
quartz vein or "Knauern") for which one mineral
assemblage is not sufficient for P-T determination with a conventional
thermobarometry study. Mineral pairs used are quartz-kyanite and
quartz-andalusite. A comparison has been done with three other surrounding
rocks with quartz-kyanite
and
quartz-garnet
pairs.
|
In both cases (tension
gashes and surrounding rocks), the pair quartz-aluminosilicates give
temperatures
comparable to these determined for metamorphic peak of temperature (600-650°C).
At such temperature and a pressure higher
than
3 kb, the presence of andalusite is doubtful. Thus this mineral
has probably been formed lately during the retrograde*
path (polymorphic*
transformation kyanite => andalusite, with conservation of isotopic ratio,
complete discussion in our publication [reference]).
Finally, the pair quartz-garnet give temperatures
of about 500-540 °C, which can be interpreted as point of mineral
formation or an average between the beginning and the end of crystallization. |

Results of temperatures obtained by oxygen isotope thermometry
(Method,
calibration : Sharp 1992, 1995) |
1) Introduction | 2) Structural | 3) THERMOBAROMETRY | 4) Tension gashes | 5) Cartography | Bibliography | Thanks
Aluminosilicate-bearing veines: 4.1) Mineralogy | 4.2) Structure | 4.3) Examples | 4.4) Formation
|
|




